Utilization of the inherent CO2-binding capacity of concrete - a contributor in the future construction industry
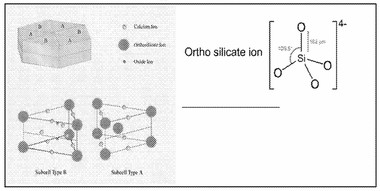
Carbonation of concrete normally occurs when air or water-borne CO2 dissolves in the concrete pore water and react with Ca2+ to form stable CaCO3. Upon carbonation, the pH of the concrete pore water is decreased to around 9. Carbonation mainly involves decalcification of the Ca-bearing hydrate phases when different polymorphs of CaCO3 are formed. In addition, the Mg-bearing hydrate phases (OH-hydrotalcite and CO3-hydrotalcite) will also carbonate by forming MgCO3 and Al(OH)3. Carbonation starts at the surface of the concrete and evolves inwards as a result of the diffusion of air and moisture. The carbonation depth is normally determined with an aqueous pH-indicator of phenolphthalein or thymolphthalein which become colorless at a pH below 10. The indicator is usually sprayed on the surface of a newly split concrete sampled (e.g. core drilled sample) and the carbonation depth (colorless area) is measured. However, the indicator cannot give an exact amount of the carbonation degree within the carbonated zone.
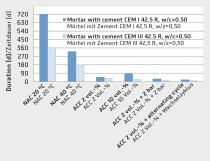
Model calculations of the CO2-binding to cementitious material containing cement has been conducted (e.g. 330 kg of CO2 per kg of Portland cement), which can be applied to concrete materials with known cement contents. A new method has been developed for cement-based materials to measure the CO2-binding more precisely. This method has been applied for crushed materials as well as for block products. It contains chambers that are determining the consumption of CO2 by the exposed materials. The temperature, moisture and CO2 concentration are controlled throughout the exposure. The new method contributes to a more precise calculation of the global warming potential in life cycle analysis for cement-based products, since CO2-binding caused by carbonation is currently not included.
The carbonation assessments become increasingly important, since it recently became mandatory to also include Module C and D when EPDs are prepared. This is specified in the European standard EN 15804:2012+A1:2013+A2:2019 that gives the core rules for the product category of construction products. It also allows for including carbonation when accounting for greenhouse gas emissions along with the more specific category rules for concrete and concrete products, EN 16757:2017. Module C includes demolition, recycling, stockpiling and potential disposal at landfill, whereas Module D includes the use of recycled products in various user applications (end of waste stages). These stages involve size reduction of concrete and potentially increased carbonation due to increased exposure of air to surface area.
Calculations have shown that around 70% of the carbonation potential of the Norwegian concrete mass (on average) is present at the end of a primary service life of 100 years. It therefore becomes important to design recycling applications that optimize the carbonation speed. The specific guidelines for how Module C and D can be documented, are currently under preparation. Preliminary calculations have shown that recycling concrete into recycled aggregates, will have a positive impact on concrete EPD. The impact on the global warming potential is attributed to decreased use of crushed natural rock materials and CO2-binding by carbonation.